
This English chemist attempted an experiment in which he heated a mineral sample with potassium carbonate. That special day in 1801, Hatchet was analysing the aforementioned mineral sample obtained from the British Museum. Originating from North America, the mineral sample was labeled columbite, in honor of the American continent discoverer, Cristopher Columbus. At the time, Winthorp was the first governor of Connecticut, United States. More than a century before Hatchett’s revolutionary chemical trial, the mineral sample that was an object of scientific interest to the English chemist was taken from a type of American ore and sent to England by John Winthrop the Younger. By examining the mineral samples exposed in the British Museum, Hatchett’s interest was caught by a particular sample for which he believed contains a new, undiscovered substance. The English mineralogist and analytical chemist Charles Hatchett (1765 – 1847) had performed numerous analytical researches on many types of minerals and ores in his private laboratory. The thickness of its oxide layer determines the color of the metal niobium, which can vary from white to blue, yellow, and green. Being corrosion-resistant, niobium metal starts to develop the protective oxide film at a temperature of 200oC. At extremely low (cryogenic) temperatures, this transition metal adopts superconducting properties in the presence of strong electric currents and magnetic fields. Niobium’s atomic radius according to van der Waals is 0.143 nm, while its electronegativity according to Pauling is unknown.Įlement 41 has a cubic crystalline body-centered structure and 2, 3, or 5 valence electrons. Despite the high melting point, niobium has a lower density than the other refractory metals. It reaches its boiling point at 4741☌, 8566☏, 5014K, while the melting point is achieved at 2477☌, 4491☏, 2750K. With the periodic table symbol Nb, atomic number 41, atomic mass of 92.91 g.mol -1, and electron configuration 4d 4 5s 1, niobium is soft and ductile paramagnetic metal. Lustrous white also commonly found in blue, green, and yellowįrom less than 44 nanoseconds to 20.300 years The symbol in the periodic table of elements Niobium is considered a technology-critical element.Chemical and Physical Properties of Niobium Property In the last two applications, the low toxicity and iridescence produced by anodization are highly desired properties. 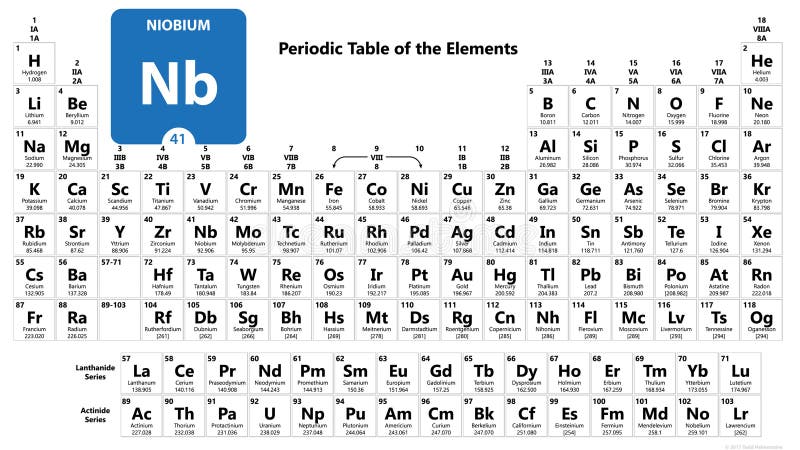
Other applications of niobium include welding, nuclear industries, electronics, optics, numismatics, and jewelry. These alloys, also containing titanium and tin, are widely used in the superconducting magnets of MRI scanners. Niobium is used in various superconducting materials. The temperature stability of niobium-containing superalloys is important for its use in jet and rocket engines. 1%, the small percentage of niobium enhances the strength of the steel by scavenging carbide and nitride. Although these alloys contain a maximum of 0. Niobium is used mostly in alloys, the largest part in special steel such as that used in gas pipelines. Brazil is the leading producer of niobium and ferroniobium, an alloy of 60–70% niobium with iron. Niobium is an important addition to high-strength low-alloy steels. It was not until the early 20th century that niobium was first used commercially. 
Niobium was officially adopted as the name of the element in 1949, but the name columbium remains in current use in metallurgy in the United States. In 18, a series of scientific findings clarified that niobium and columbium were the same element (as distinguished from tantalum), and for a century both names were used interchangeably. German chemist Heinrich Rose determined in 1846 that tantalum ores contain a second element, which he named niobium. 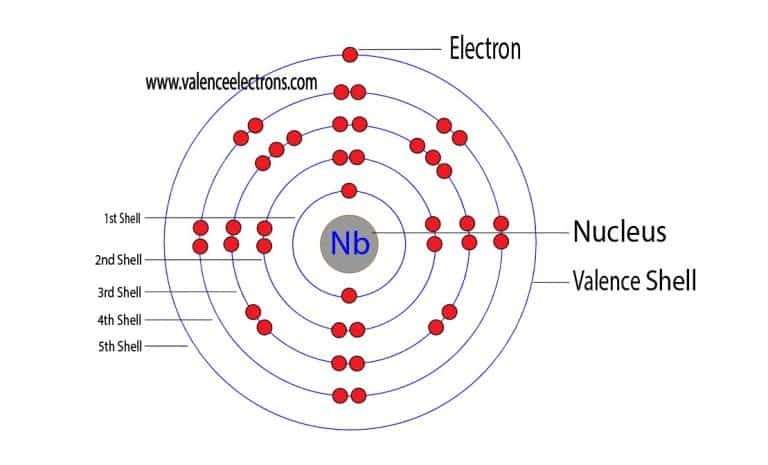
In 1809, English chemist William Hyde Wollaston wrongly concluded that tantalum and columbium were identical. English chemist Charles Hatchett reported a new element similar to tantalum in 1801 and named it columbium. The name reflects the great similarity between the two elements in their physical and chemical properties, which makes them difficult to distinguish. Its name comes from Greek mythology: Niobe, daughter of Tantalus, the namesake of tantalum. Niobium is often found in the minerals pyrochlore and columbite, hence the former name "columbium". Niobium oxidizes in Earth's atmosphere very slowly, hence its application in jewelry as a hypoallergenic alternative to nickel. Pure niobium has a Mohs hardness rating similar to pure titanium, and it has similar ductility to iron. It is a light grey, crystalline, and ductile transition metal. Niobium is a chemical element with chemical symbol Nb (formerly columbium, Cb) and atomic number 41.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
